The impact of genetics, stress, and lifestyle: Q & A with Eric Rimm
Every heart attack survivor has a story to tell. Harvard School of Public Health Leadership Council member Rick Smith’s is a tale of good fortune.
There is no history of heart disease in Smith’s family. Slim, athletic, and health-conscious at age 56, he exercises regularly and has always eaten healthfully. He doesn’t smoke. Stress, however, has for many years been part of Smith’s job in New York City’s investment industry.
Smith had a video X-ray of his heart called a CT angiogram taken in 2006, but only because a former Harvard College roommate insisted he do so. To Smith’s astonishment, his calcium score—a measure of atherosclerosis that he presumed would be normal, meaning 100 or less-was 2,500. A few of his coronary arteries were 60 to 80 percent blocked.
Bypass surgery helped save Smith’s life in February of 2008. In October, he and his wife, Charlotte, asked HSPH Associate Professor Eric Rimm, director of the Program in Cardiovascular Epidemiology, to talk with other HSPH friends and supporters about factors useful for predicting heart disease risk-some well known, others less so. Cardiovascular disease is the number one cause of premature death in the United States, killing about 870,000 people a year. Of these, female victims outnumber males by roughly 50,000, partly because their symptoms too often go unrecognized. What’s the average person to do? Highlights from Rimm’s talk offer answers.
Q: How can the average American ward off heart disease?
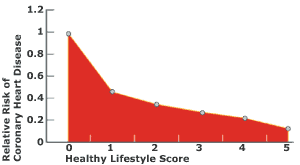
A: Our research tells us that Americans’ heart attack risk can be cut by more than 80 percent by adopting five lifestyle factors: not smoking, a healthy diet, maintaining a healthy weight, regular physical activity, and light to moderate alcohol consumption.
Some people may benefit from medications-a statin to lower blood cholesterol, say, or a blood-pressure drug. But a healthier lifestyle is even more critical for these people, because they’re at highest risk.
Q: What about stress? Can it really cause heart disease?
A: Yes, stress can lead to heart disease. For example, it can disrupt cardiac function, either by altering the electrical stability of the heart or by hastening atherosclerosis and increasing systemic inflammation. All of these processes increase the likelihood of a heart attack.
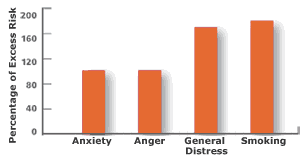
Though hard evidence is only now emerging, anxiety, anger, and other forms of distress appear to add to our risk of heart disease. These different types of distress can be measured using rigorous methods and then studied in relation to risk of developing heart disease (see bar chart).
Obviously, it’s a huge challenge to collect accurate measures of stress, especially given the wide range of people’s responses. My colleague Laura Kubzansky, an associate professor of Society, Human Development, and Health, is working on this now, and also looking at how stress and emotion impact health. The simplest measures ask people about their feelings and include statements covering a range of intensity of feeling. For example, a measure of anxiety might include statements from “I worry quite a bit over possible misfortunes” to “I frequently find myself worrying about something” to “I sometimes feel that I am about to go to pieces.” Answers put people somewhere on a scale ranging from “not very” to “extremely” anxious.
Under stress, we react with a set of biological responses designed to prepare the body to cope with an emergency. These responses are often adaptive in the short-term, allowing us to respond effectively to immediate challenges. However, if these challenges occur too frequently, or if we are unable to turn off the stress response, these biological responses can lead to a kind of physiological wear and tear, and health problems can arise.
Evidence so far suggests that stress may be involved not only in cases where people have underlying or overt cardiovascular disease, but also in the development of disease. As both animal and human studies have shown, stress is inversely linked to longevity.
We don’t yet know exactly how much stress it takes to cause specific health problems, or at what point over the life course stress is most damaging. Recent research suggests that distress in childhood predicts both adult levels of distress 30 years later and a variety of physical health outcomes as well. If we live to age 50, discover yoga, and are stress-free for the rest of our lives, will we be able to rid ourselves of accumulated damage from stress? We don’t yet know, but this is something investigators, including Laura Kubzansky, are exploring.
Impact of Healthy Lifestyle on Coronary Heart Disease RiskThis chart shows the relative risk of heart disease in U.S. men depending on how many of the five most influential healthy lifestyle behaviors (i.e., not smoking, healthy diet, maintaining a healthy weight, regular physical activity, and light to moderate alcohol consumption) they report employing. The healthy lifestyle score on the horizontal axis ranks men from low to high on a scale of 0 to 5, where 5 = individuals who report employing all five of the healthy behaviors. A man who performs all of these healthy behaviors is 80 percent less likely to develop heart disease than a man who performs none of them. Those who change their lifestyle by adopting even one healthy behavior can cut their risk of heart disease by more than 50 percent. Source: Health Professionals Follow-up Study, 1986–2004, Circulation |
Q: If stress and lifestyle might not fully explain heart disease in people like Rick Smith, what about genetics?
A: The human genome-our full complement of DNA, about 22,500 genes-is an important focus for the future. At points all along our DNA there are tiny, very subtle variations that make people different. Most “gene variants” have no apparent effect on health, but a few can heighten susceptibility to a disease, such as cardiovascular disease.
Using high-powered gene-reading technology and a tube of blood from each of our study participants, we can compare DNA from people with heart disease to DNA from a healthy group, and ask: Which of these tiny points of difference-technically, they’re called “single nucleotide polymorphisms,” or SNPs (“snips”)-correlate with heart disease risk? Is risk affected whether we’ve inherited a particular SNP from one parent or both?
Two years ago, you could only compare about 100,000 SNPs in a large group of people. Now you can look at a million. Next year it will be more.
Even a SNP that’s quite rare, yet still significantly more common among heart disease patients than healthy people, might clue us in to biological pathways leading to disease. We know from genome scans that a single SNP leads some people to have freckles, or eye disease, or breast cancer. Now we can look for SNPs for diabetes, celiac disease—even a condition as complex as cardiovascular disease, where many genes may be involved.
Q: That sounds mind-bogglingly complicated.
A: This work requires immense computer power and statistical expertise. Merck, the drug company, assists us with data processing. From our ongoing studies of 300,000 men and women, we’ll take 1,000 who have had a heart attack, along with their million SNPs, and another still-healthy 2,000, and their million SNPs. And we’ll see which genes are “talking together” to increase heart disease risk.
What’s so novel about our work is that we can combine all this information on genetics with 30 years of data we’ve collected on people’s diet and lifestyle in middle and older ages. We hope to learn about health factors that can modify a genetic predisposition to coronary disease.
Q: For someone who has a history of heart disease, or major risk factors, what’s next?
A: A lot of people see their physician, get a prescription, and stop there. Big mistake! In one study that looked at men who take medication, we still found lifestyle to be critically important. A “medications only” group had hundreds more heart attacks compared to the healthy-lifestyle group. Medication only cuts heart attack risk by 30 percent. It’s not enough.
Impact of Stress on Heart Disease RiskRisk of heart disease increases as levels of anxiety, anger, or more general symptoms of distress increase. These effects have been observed for both men and women. The increase in risk due to distress (roughly 170 percent) is not much less than that of smoking (about 180 percent), which is already a powerful and well-documented risk factor. Sources: L. Kubzansky, Cleveland Clinic Journal of Medicine, 2007; S. Yusuf et al., Lancet, 2004 |
Q: What about diet?
A: We think the Mediterranean diet will be the healthiest option. You want a healthful diet that you can stick to for a lifetime, not just weeks or months. Rather than worry about the total amount of fat you eat, worry about the type. Aim for ‘good’ monounsaturated and polyunsaturated fats, such as olive oil and other vegetable oils. Avoid trans fats. Limit saturated fats; choose lean proteins. Go for whole grains and other minimally processed carbohydrates.
My HSPH colleague Meir Stampfer and his collaborators recently published the longest study to date in which people testing three contrasting diets actually adhered to them closely: a low-fat diet of less than 30 percent fat (10 percent saturated), 20 percent protein, 50 percent carbohydrate; a Mediterranean diet of up to 35 percent fat (including olive oil and nuts), 50 percent carbohydrate, the rest protein; and the Atkins diet of about 40 percent fat, 20 percent protein, and the rest carbohydrate.
This study was conducted in Israel at a company where participants were fed lunch, the main meal in that country. Compliance rates were 85 to 90 percent over two years, almost twice as high as rates typically achieved in U.S. studies.
In the beginning, all three diets led to weight loss. But people couldn’t stick with the low-fat diet-they were too hungry. In the long run, they lost the least amount of weight. Given its links to lower heart disease and colon cancer risk, we recommend the Mediterranean diet.
Q: What’s the story with fish? Good or bad?
A: Far too much media attention is paid to contaminants in fish, which in no way outweigh its health benefits. Recently a colleague and I pulled together the world’s literature on fish and heart disease, and on fish and death rates from any cause. If you ate two servings of fish a week, depending on the type, you’d take in 250 to 1,000 mg of fish oil containing heart-healthy omega-3 fatty acids on average, per day. Among those eating no fish, there is a substantially higher risk for sudden death by coronary heart disease than among people eating as little as 250 mg of fish oil a day. The biggest benefits come from just one or two servings of salmon a week.
Q: How should someone eat who’s already had a heart attack?
A: There is really only one large-scale, long-term trial, in men, where they randomized heart-attack survivors to a low-fat diet or to a Mediterranean diet. In terms of survival, men on the Mediterranean diet did much better.
That goes to show that dietary fat is actually good for you, so long as it’s mostly polyunsaturated fat (including omega-3 fats) and monounsaturated fats.
Q: What sets HSPH apart from other research institutions?
A: Here, scientists working with animals are interacting with epidemiologists working with human populations. That speeds up advances.
For instance, my HSPH colleague Gökhan Hotamisligil, chair of the Department of Molecular Metabolism, uncovered the function of a gene that essentially prevents diabetes in mice. He found that, when you breed mice without this gene, called Ap2, you can feed the mice lots of fatty foods and they still won’t get the high blood sugar and other clinical warning signs for diabetes that you’d expect in a typical mouse. We can’t delete people’s genes the way we do by breeding mice, but a drug could have the same effect.
Q: How is HSPH’s mouse discovery relevant to humans?
A: We sequenced the comparable gene for Ap2 in humans and found that about 3.5 percent of the population has one copy of a rare SNP that may somehow change the gene’s function. In about 8,000 people, women with this SNP had about 50 percent lower risk of a heart attack. Men had about 27 percent. Overall, the SNP was linked to a one-third lower risk in humans. Now, pharmaceutical companies and university scientists are looking to block or limit Ap2’s protein production.
We can now measure the Ap2 protein in blood. Our study participants with high Ap2 levels had a 50-percent higher risk of heart disease, even after accounting for red flags for risk like cholesterol and inflammatory molecules.
It’s rewarding to apply lab discoveries to humans. This initial Ap2 work was done one SNP at a time for one gene. But in the body, proteins made from multiple genes are all “talking” and signaling one other at once.
Heart disease, obesity, and cancer likely involve many SNPs and genes. To study links between a million SNPs and health gets challenging. But computer algorithms based on our biological knowledge should help us understand the complex origins of chronic diseases. It’s incredibly exciting.
Q: What resources do you need?
A: In the last five years, government funding rates have dropped-especially for new research. Faculty salaries largely come out of those grants. We’ll need financial resources to bring in students and postdocs. We want to engage thousands of new study participants with broader genetic backgrounds and ages. And we’ll need computing infrastructure to support this work. Asking for support for big freezers that store 3 million blood samples isn’t very sexy. But it takes enormous resources to run a “freezer farm,” with its huge liquid nitrogen coolers. Each of these giant containers holds 18 to 27 racks holding 10 boxes of 100 tubes each.
A top priority is funding projects that pull together all our expertise at HSPH. We’re training today’s students to work across scientific disciplines. Mastering one is no longer enough.
Karin Kiewra is the associate director of Development Communications at HSPH and editor of the Review.
Photograph, Kent Dayton/HSPH