Study of lipid metabolism in neurons may offer therapeutic possibilities for neurodegenerative diseases
For immediate release: April 13, 2020
Boston, MA – Disrupting the production of a class of lipids known as sphingolipids in neurons improved symptoms of neurodegeneration and increased survival in a mouse model, according to new research led by the joint laboratory of Robert Farese, Jr. and Tobias Walther at Harvard T.H. Chan School of Public Health and Howard Hughes Medical Institute.
The findings, published online April 13, 2020 in the Proceedings of the National Academy of Sciences (PNAS), could help in the development of therapies for a range of neurodegenerative diseases.
“This work began in yeast, and serendipitously we discovered that mutations related to neurodegeneration in humans led to abnormalities in cell lipid metabolism. Most investigators don’t look at the lipids, and we were quite excited and surprised,” said Walther, professor of molecular metabolism and executive director of the Harvard Chan Advanced Multi-omics Platform.
In the study, the Farese & Walther Laboratory identified a link between sphingolipid metabolism and a mutation that affected vesicle trafficking, the process by which molecules are transported to different parts of a cell. Defects in the trafficking process are known to play a role in neurodegenerative diseases, but the exact mechanism of the effect is not understood.
The Farese & Walther Laboratory has previously focused their research efforts on the Golgi-associated retrograde protein (GARP) complex. Earlier studies in yeast and flies showed that mutations in GARP proteins lead to sphingolipid abnormalities and stunted cell growth, and that these effects could be reversed by inhibiting sphingolipid metabolism. Many neurological diseases are caused by mutations in genes that are involved in lipid metabolism, and changes in lipid metabolism have been reported in amyotrophic lateral sclerosis (ALS), Parkinson’s disease, and Alzheimer’s disease. With that in mind, the Farese & Walther Laboratory investigated whether modulating sphingolipid metabolism affects cell dysfunction as it relates to neurodegenerative diseases.
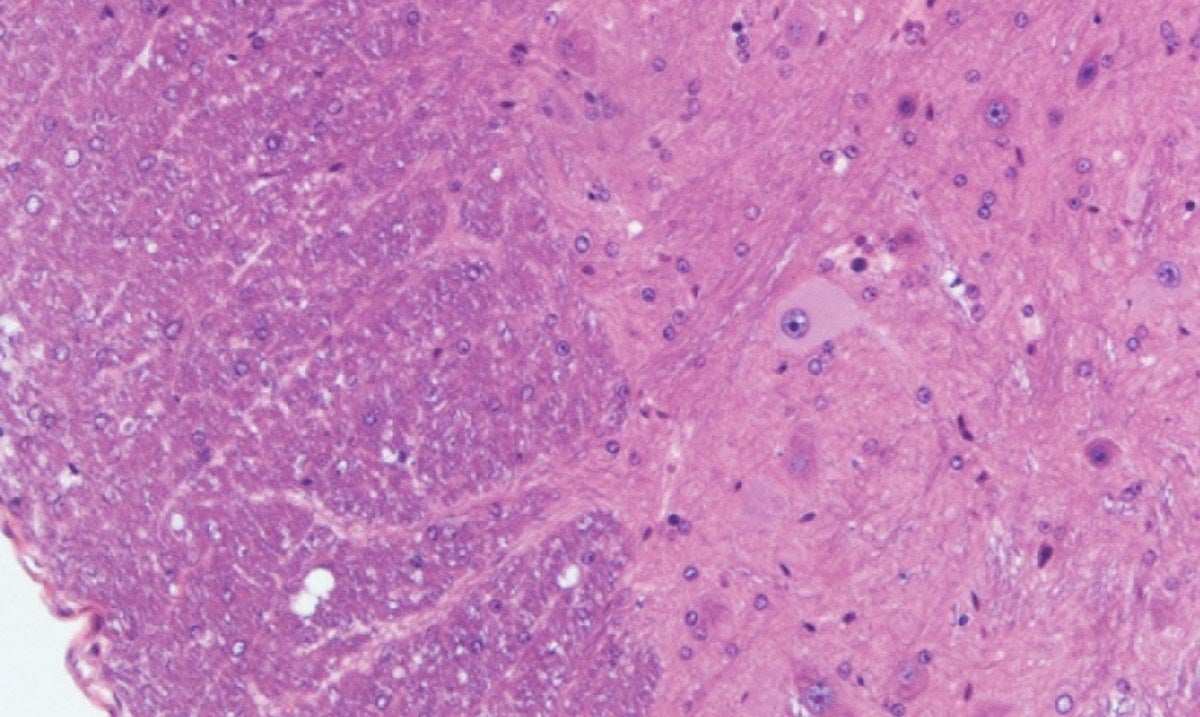
The research team, which included Harvard Chan scientists Constance Petit and Jane Lee, used a model known as wobbler mice, which have a mutation in a specific GARP protein called VSP54. That mutation causes motor-neuron disease similar to ALS. They found that sphingolipid molecules that were toxic to cells accumulated in spinal cords of wobbler mice, as well as in embryonic fibroblasts, a type of cell that was cultured from the mice. Furthermore, they showed that certain molecules involved in sphingolipid metabolism were misplaced in neurons from wobbler mice.
The team then found that treating the mice with the sphingolipid synthesis inhibitor myriocin, which is already used as an antifungal and immunsuppreive drug, prevented the buildup of sphingolipids, reduced their toxic effects, improved wellness scores in the mice, and ultimately extended the animals’ lifespan.
The results indicate that compromised sphingolipid metabolism in GARP mutationsare a potential cause of neurodegenerationand that correcting defects in sphingolipid metabolism might restore neuronal function. Sphingolipid metabolism may therefore be an important target for therapeutic development for neurological disorders associated with mutations in membrane trafficking.
“We are cautiously optimistic,” Farese said. “Perhaps targeting lipid abnormalities provides a new therapeutic angle for some diseases of neurodegeneration.”
Other Harvard Chan School researchers who contributed to the work include Sebastian Boland, Romain Christiano, Zon Weng Lai, Niklas Mejhert, Shane Elliott, and Sara Haque. Researchers from Harvard Medical School, the Broad Institute of MIT and Harvard, and the University of California San Francisco also contributed to the study.
The work was supported by the Consortium for Frontotemporal Dementia Research, the Mathers foundation, National Institutes of Health grant R37NS083524, the Harvard Brain Initiative ALS seed grant program, the Canadian Institutes for Health Research, and the Howard Hughes Medical Institute.
“Inhibition of Sphingolipid Synthesis Improves Outcomes and Survival in GARP-Mutant wobbler Mice, a Model of Motor-Neuron Degeneration,” Constance S. Petit, Jane J. Lee, Sebastian Boland, Sharan Swarup, Romain Christiano, Zon Weng Lai, Niklas Mejhert, Shane D. Elliott, David McFall, Sara Haque, Eric Huang,Roderick T. Bronson, J. Wade Harper, Robert V. Farese, Jr. and Tobias C. Walther, Proceedings of the National Academy of Sciences, online April 13, 20 doi: 2010.1073/pnas.1913956117
photo: The Farese & Walther Laboratory
Visit the Harvard Chan School website for the latest news, press releases, and multimedia offerings.
For more information:
Chris Sweeney
617.432.8416
csweeney@hsph.harvard.edu
###
Harvard T.H. Chan School of Public Health brings together dedicated experts from many disciplines to educate new generations of global health leaders and produce powerful ideas that improve the lives and health of people everywhere. As a community of leading scientists, educators, and students, we work together to take innovative ideas from the laboratory to people’s lives—not only making scientific breakthroughs, but also working to change individual behaviors, public policies, and health care practices. Each year, more than 400 faculty members at Harvard Chan School teach 1,000-plus full-time students from around the world and train thousands more through online and executive education courses. Founded in 1913 as the Harvard-MIT School of Health Officers, the School is recognized as America’s oldest professional training program in public health.


