Manning Lab
The Manning lab is defining the molecular connections between nutrient signaling and metabolic networks under both physiological and pathological states. Lab researchers are particularly focused on the role of the PI3K-mTOR signaling network in the control of cellular and systemic metabolism and its impact on the insulin response, type-2 diabetes, cancer, neurological disorders, and aging.
665 Huntington Ave
Building 2, Rm. 127
Boston, MA 02115
Welcome to the Manning Lab
Led by Brendan D. Manning, Ph.D., the lab is focused on defining the regulation and function of the PI3K-mTOR signaling network under both physiological and pathological settings.
To do so, a multi-tiered program of hypothesis-driven approaches utilizing genetics, biochemistry, cell biology, and animal models combined with unbiased systems-level tools, including genomics, proteomics, metabolomics, and bioinformatics, is employed.
The lab’s research aims are based on the premise that delineating the complex circuitry of signaling networks within cells will provide insight into how best to directly stimulate or inhibit these pathways, as appropriate, for the prevention or treatment of human diseases, including cancer and type-2 diabetes.
Our Research
Research in the Manning Lab is focused on unraveling the key regulatory circuits that control cell growth. They are delineating how signals from nutrients and growth factors are propagated to coordinately regulate metabolism, with implications in a wide variety of complex human diseases. Research efforts are defining the molecular control mechanisms and functions of the ubiquitous PI3K-mTOR signaling network, which senses and relays signals from nutrients and other growth cues to alter key metabolic processes in cells and tissues.
Frequent dysregulation of the PI3K-mTOR network contributes to a diverse set of seemingly unrelated human diseases, including those with uncontrolled cell growth and metabolism as an underlying feature. This network is aberrantly regulated in, or believed to contribute to, the majority of human cancers, genetic tumor syndromes (e.g, tuberous sclerosis complex, lymphangioleiomyomatosis, PTEN syndrome), metabolic diseases (e.g., obesity, diabetes, cardiovascular disease), autoimmune and inflammatory diseases (e.g., arthritis, lupus, hepatitis), and neurological disorders (e.g., epilepsy, autism, neurodegeneration).
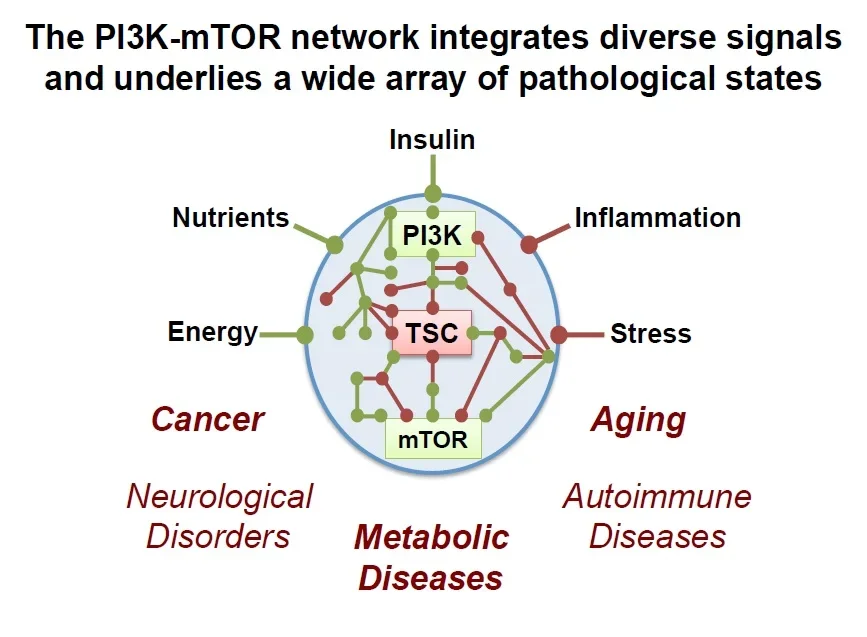
This signaling network also influences the lifespan of organisms and serves as a major connection between diet and the aging process. The Manning lab seeks to decipher the circuitry of the PI3K-mTOR network under both physiological and pathological states, and how its downstream functions contribute to metabolic homeostasis and dysfunction in human health and disease.
Cancer, we now know, is caused by the uncontrolled growth of a single cell. . . In a normal cell, powerful genetic circuits regulate cell division and cell death. In a cancer cell, these circuits have been broken, unleashing a cell that cannot stop growing. That this seemingly simple mechanism – cell growth without barriers – can lie at the heart of this grotesque and multifaceted illness is a testament to the power of cell growth.
Siddhartha Mukherjee, The Emperor of All Maladies
Areas of Research Focus
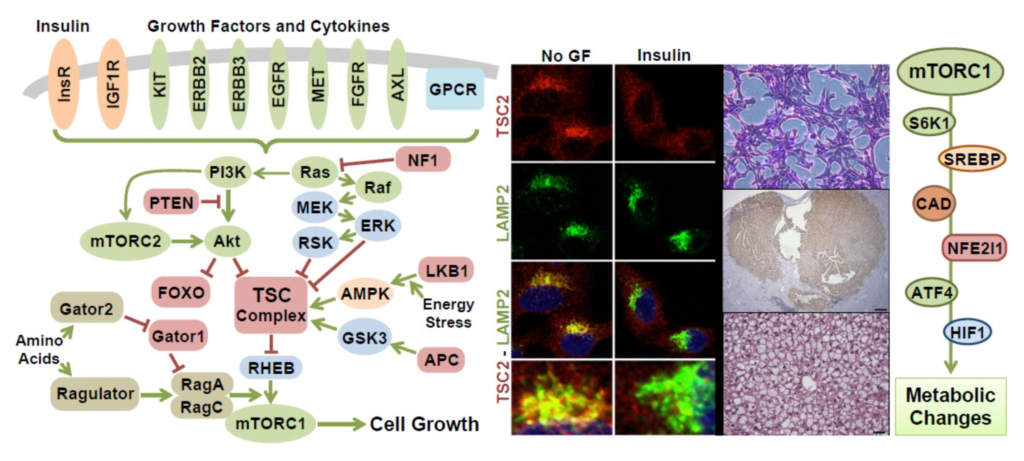
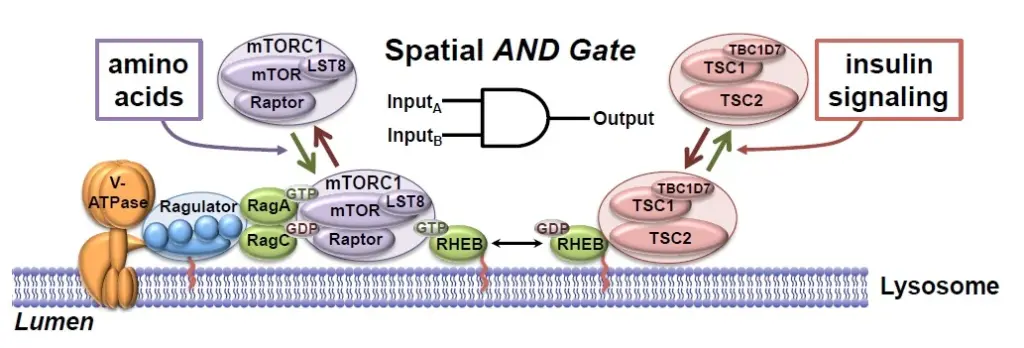
The Manning lab is delineating the molecular wiring and functions of a signal transduction network that senses and responds to nutrients and other growth stimuli, is aberrantly regulated in a diverse set of human diseases, and plays an evolutionarily conserved role in aging. In normal cells, the PI3K-mTOR signaling network is exquisitely regulated by both cell intrinsic signals, in the form of nutrients and cellular stress, and exogenous signals from secreted growth factors, cytokines, and hormones. The Manning lab has found that these physiological signals are sensed in an integrated manner that assures that the pathway is spatially and temporally activated only under optimal local and systemic nutrient conditions. Several projects in the lab are aimed at defining regulatory mechanisms involving the TSC protein complex, which is a central, signal-integrating node within this vast network. Furthermore, negative feedback mechanisms are prevalent within the network and have a profound influence on the intensity and duration of signal propagation, as well as the response of the network to pharmacological interventions. The importance of a mechanistic understanding of this regulation is underscored by the fact that aberrant signaling within the network is a common feature of pathological states, including cancer, type-2 diabetes, and obesity.
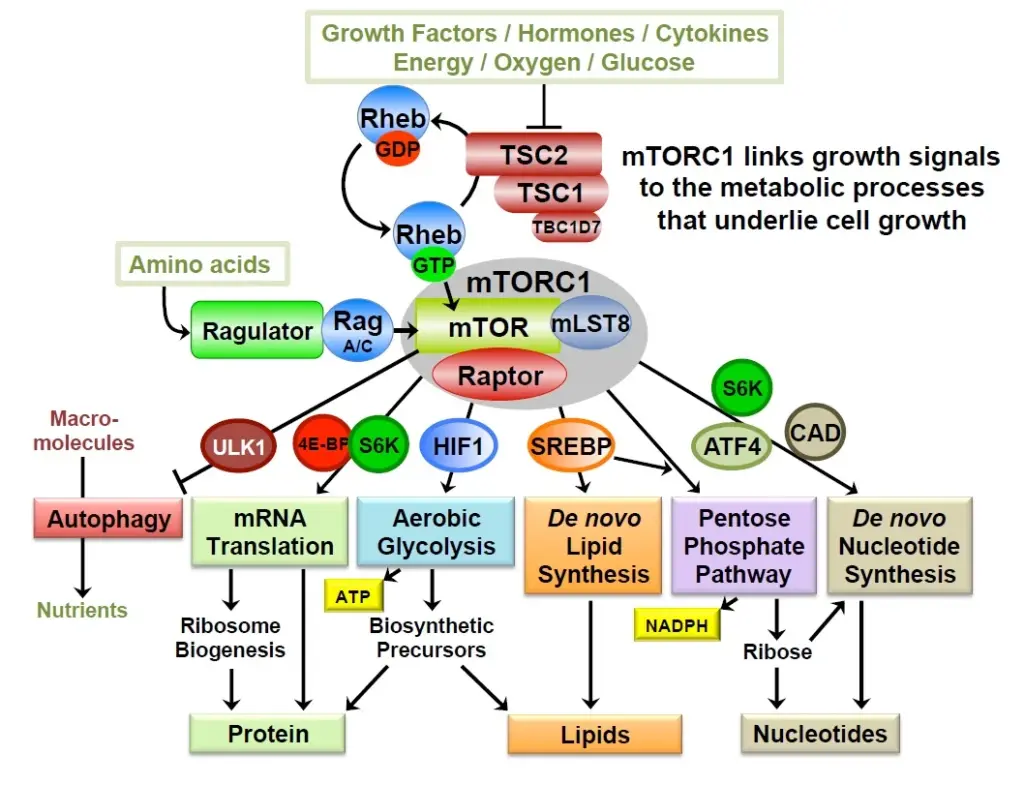
In studies initiated through a variety of unbiased omics approaches, the Manning lab has found that the primary function of the PI3K-mTOR network is to control cellular metabolism. The network serves to link growth signals to mechanisms controlling the key anabolic processes that underlie growth. Activation of mTOR complex 1 (mTORC1) in response to nutrients and other growth signals promotes the conversion of nutrients into biomass through downstream transcriptional and post-translational effects on major metabolic enzymes, such as those influencing protein, lipid and nucleotide synthesis. Through these molecular mechanisms, the network controls cell and tissue growth, as well as integrated systemic metabolism. The lab is particularly interested in cancer metabolism and how the common dysregulation of PI3K and mTORC1 in human tumors alters the metabolism of tumor cells and distinguishes them from their cells of origin.
The Manning lab is defining the mechanisms and consequences of PI3K-mTOR network dysfunction in a variety of pathological states. One such setting is the neurological and tumor syndrome tuberous sclerosis complex (TSC), as well as the related proliferative lung disorder lymphangioleiomyomatosis (LAM). TSC and LAM are caused by inactivating mutations in TSC1 or TSC2, which are tumor suppressors that form a protein complex (the TSC complex) that is a central negative regulator of mTORC1. In addition to serving as the key link between growth factor signaling and mTORC1 activation, the TSC complex is also a shared downstream target of the most common oncogenic signaling pathways, including PI3K and RAS, resulting in aberrant regulation of the TSC complex and activation of mTORC1 in the majority of sporadic cancers. Through mechanisms that are currently unknown, exquisite control of signaling through the PI3K-mTOR network is also perturbed by nutrient excess and obesity, as well as over the course of organismal aging. In a context-dependent manner, these signaling defects can contribute to metabolic reprogramming in cancer cells and the development of insulin resistance and metabolic dysfunction underlying type-2 diabetes. These are areas of active investigation in the lab.