[ Winter 2008 ]
Scientists trace inflammatory ulcerative colitis to failure of immune system “peacekeeper”
A delicate barrier is all that stands between billions of foreign bacteria living in the large intestine, or colon, and the misery of ulcerative colitis. This chronic, poorly understood disease afflicts a half-million people in the United States and as many as four million worldwide.
If hosting a vast and motley population of alien bugs inside the colon would seem to be asking for trouble, recall from high school biology that humans rely on these microbes for digestion. Evolution has crafted a means of peaceful co-existence: an intestinal demilitarized zone made up of the colon’s single-celled lining and vigilant “peacekeeper” molecules inside the colon wall that restrain the body’s immune defenses.
Amazingly, this system works well most of the time. But in some people, for reasons long mysterious, the truce turns sour. Intestinal bacteria become nasty, gaps open in the colon lining, and defender immune cells go on the attack. The result is a wave of inflammation that creates open sores, or ulcers, up and down the colon, fomenting pain, frequent diarrhea, nausea, and bleeding, which can become a lifetime burden. Anti-inflammatory drugs can partially alleviate symptoms, but in the worst cases, surgeons often remove the entire colon, also known as the large bowel.
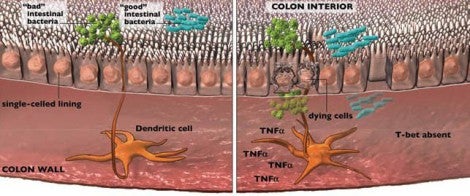
In a surprising discovery, HSPH immunologist Laurie Glimcher and postdoctoral fellow Wendy Garrett and their team have unmasked the immune peacekeeper in mice and showed how its absence triggers the inflammatory border conflict. What’s more, after finding that mice with ulcerative colitis can pass their disease on by transmitting gut bacteria, they cured those cases with standard antibiotics.
Inflammatory bowel disease: A short courseInflammatory bowel diseases, which include ulcerative colitis and Crohn’s disease, affect 1.4 million in the U.S. About 30,000 new cases are diagnosed each year, chiefly in people ages 15 to 35. Ulcerative colitis involves only the large intestine, whereas Crohn’s can develop anywhere in the gastrointestinal tract, from the anus to the mouth. Although scientists and physicians have long blamed a faulty immune system, inheritable DNA changes are suspected, since the disease begins so early in life. Both conditions can affect cells of the skin, bones, joints, and eyes. Persistent diarrhea, cramps, fever, rectal bleeding, weight loss, and fatigue are typical and may be intermittent. Symptoms vary and are unpredictable. The goal of available treatment is to ease symptoms and achieve remission. Drugs include aspirin-like compounds, corticosteroids, immune modulators, antibiotics (for Crohn’s), and biological therapies, such as antibodies that block TNF-alpha. Removing the entire large bowel eliminates ulcerative colitis. Surgery can help Crohn’s patients, but it isn’t a cure. |
Like mouse, like human
In the October 5 issue of Cell, the investigators reported that mice lacking the peacekeeper molecule—a protein called T-bet made in immune dendritic cells—developed an aggressive disease that mimics ulcerative colitis in humans. So close is the resemblance, in fact, that the mice represent an invaluable model for researchers searching for new and better human treatments. Indeed, humans possess a gene-protein T-bet nearly identical to that of mice.
“That was really quite a surprise to us,” says Glimcher, the Irene Heinz Given Professor of Immunology at HSPH and Professor of Medicine at Harvard Medical School. “We didn’t expect T-bet would be a new player in the very delicately balanced regulatory system that’s necessary for keeping the intestinal wall intact and healthy.”
Equally surprising was that diseased mice could transmit their illness to mice whose immune systems were normal, their T-bet function undisturbed. Successfully blocking transmission of their colitis with antibiotics proved that immune malfunction had led to the production of harmful bacteria and was indeed the key to this perplexing disease.
The road to T-bet
Basic research often leads to serendipitous findings, as the HSPH team’s discovery goes to show. Several years ago, Glimcher was not on the hunt for the cause of ulcerative colitis at all. Rather, her lab was studying mice genetically engineered to lack T-bet, a regulatory protein “transcription factor” that she and a former postdoctoral fellow, Suzanne Szabo, had isolated in 2000.
T-bet turned out to be the long-sought “switch” that directed immature T-cells to become mature TH1 cells rather than TH2 cells; the two types have differing roles in immunity. Subsequently, her group and others showed T-bet to play a role in both tolerance—restraining inappropriate immune responses to “self” proteins—and failed tolerance, which underlies autoimmune diseases and allergies.

Then, three years ago, the lab’s T-bet knockout mice started dying off. Glimcher asked Garrett to investigate. Many of the sick mice had inflamed colons, “and when we see that, we immediately think of inflammatory bowel disease,” Garrett recounts.
“All of these mice lacked T-bet-and by four weeks of age, 100 percent of them developed the disease,” Glimcher notes. She, Garrett, and others, including former HSPH postdoctoral fellow Graham Lord, followed this lead. They found that T-bet is normally expressed in immune dendritic cells patrolling the epithelium, which lines organs and interacts with the external environment, including the bacterial contents of the colon. But in mice that had T-bet knocked out and also lacked T and B cells, the colon’s dendritic cells cranked out too much tumor necrosis factor (TNF-alpha), a pro-inflammatory hormone-like substance. TNF-alpha injured the bowel lining in places, essentially by inducing epithelial cell death.
The stage was set for the inflammatory immune response.
A communicable disease?
It appears the T-bet protein’s job is to keep TNF-alpha in dendritic cells under control, to “maintain harmony between the host and bacteria in the gut,” Garrett explains. Lose T-bet, and bad things happen.
Without T-bet, the mix of intestinal bacteria appears to change, favoring dangerous pathogens, the investigators observed. A big surprise was that T-bet knockout mice in which ulcerative colitis developed passed the disease to offspring with normal T-bet levels, and even to unrelated healthy adult mice, presumably through oral-fecal contact.
The researchers didn’t know which types of bacteria were causing the colitis, but broad-spectrum antibiotics cured it.
That the disease could be transmitted this way “is quite a striking finding,” Garrett says. “It’s amazing to us that the loss of an immune system protein in the host changes the mix of bacteria in the colon in a way that causes disease and drives colitis in healthy mice.”
That said, Garrett emphasizes, “We are not suggesting that inflammatory bowel disease is an infectious or communicable disease in humans.” Even in poor countries where fecal contamination of water is widespread, there is no evidence to suggest that the disease spreads between people.
Using their mouse model, the HSPH researchers showed that ulcerative colitis could be cured using antibodies that block TNF-alpha. Anti-TNF drugs are already used to treat inflammatory bowel disease, but the results and safety profile are far from ideal. Perhaps, Glimcher says, it might be possible to keep TNF-alpha under control by boosting T-bet in colonic dendritic cells. Another treatment strategy, one already used by some patients, is to consume “probiotic” foods, such as yogurt, to nurture the colonies of “good” bacteria that normally keep harmful microbes from getting out of hand.
What’s Next
Do people prone to ulcerative colitis inherently lack T-bet? The scientists are pursuing this possibility in human studies. They suspect that inheritable DNA alterations, or polymorphisms, might explain impaired T-bet function in some individuals who develop the disease.
It’s worth noting that Garrett, who also does research at nearby Dana-Farber Cancer Institute, is interested in a possible link between inflammation and cancer. Does the T-bet finding have any such implications?
“We certainly are exploring what others have long observed,” Garrett says, hinting at what her next step might be. “Ulcerative colitis is among the top three risk factors for colorectal cancer.”
Richard Saltus has written about science and medicine for the Boston Globe, the Associated Press, the New York Times, the San Francisco Examiner, the Howard Hughes Medical Institute Bulletin, and other publications.