Park Lab
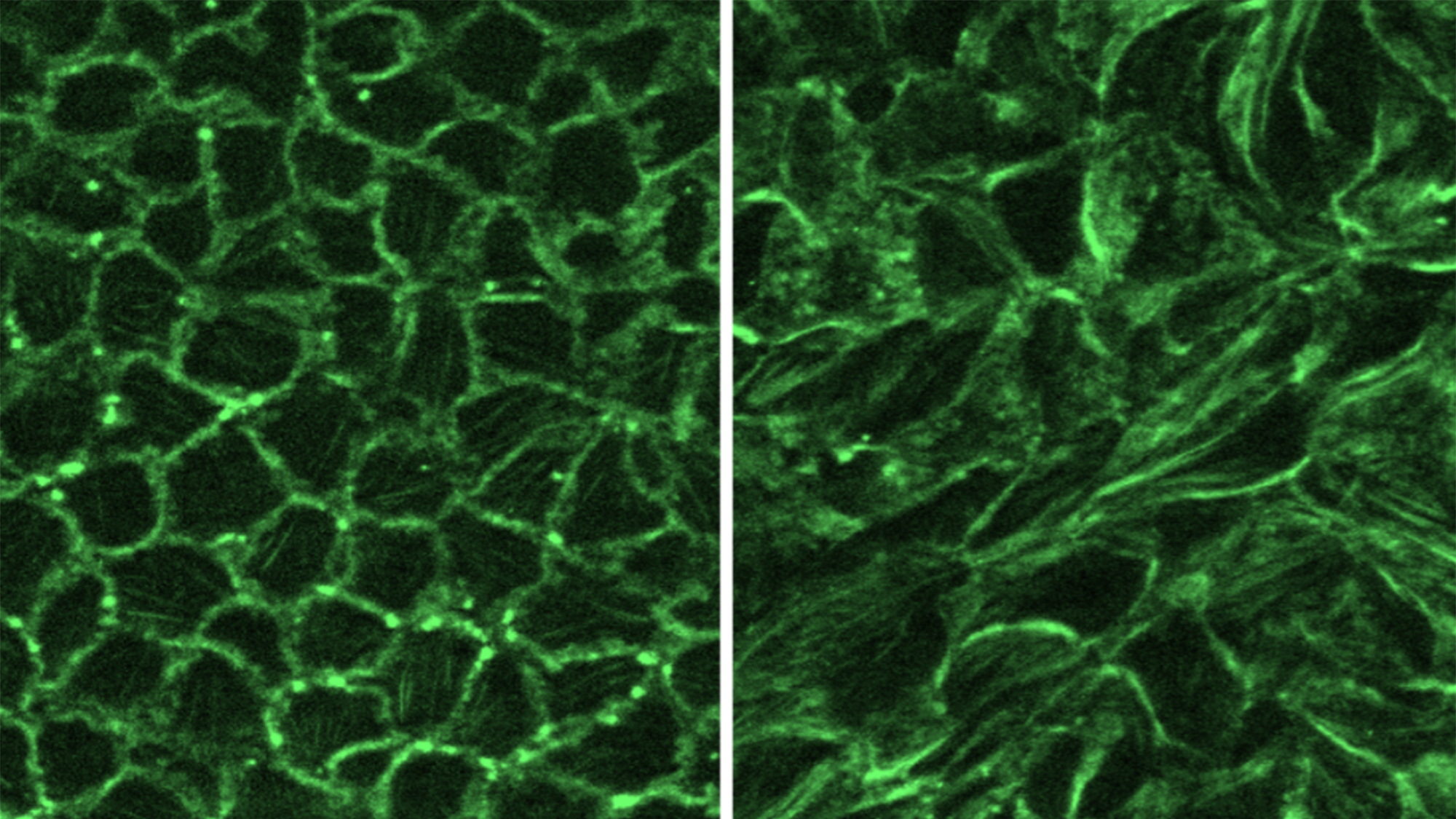
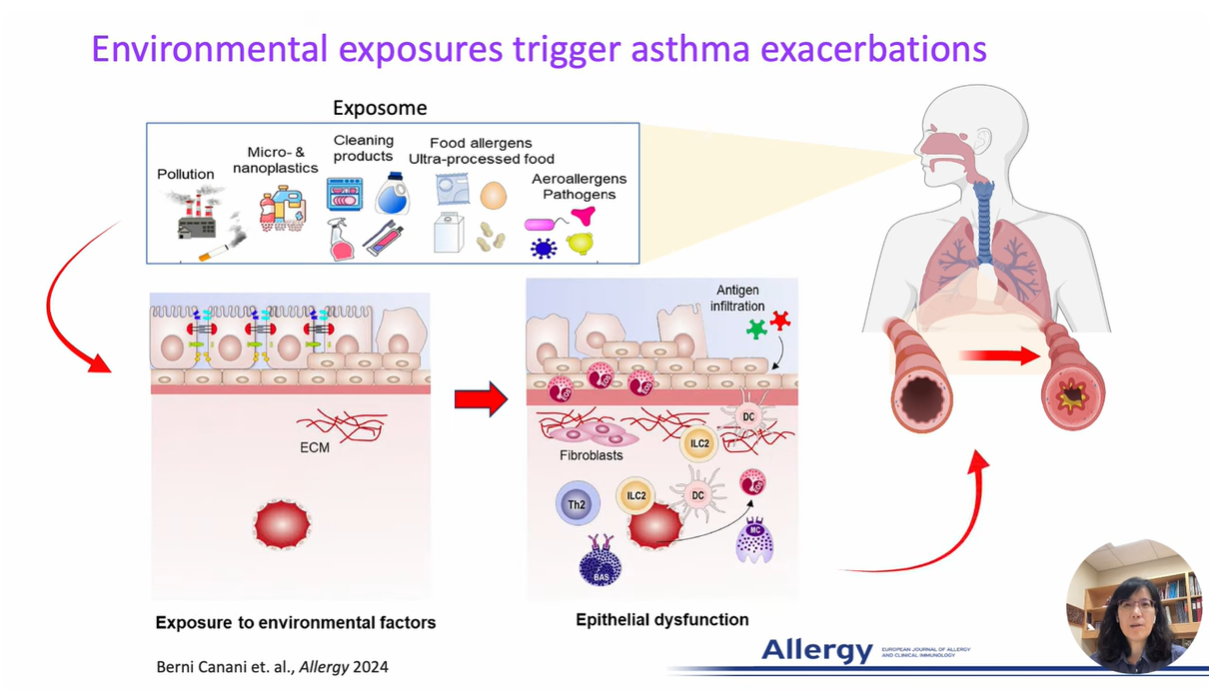
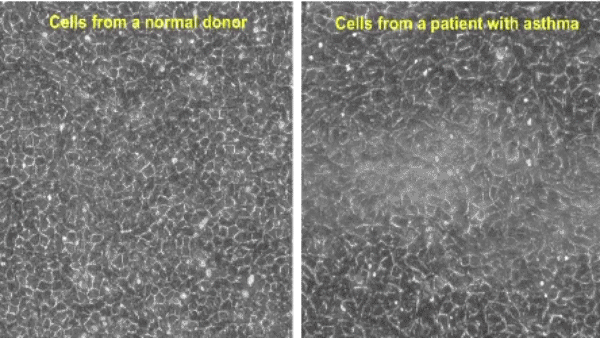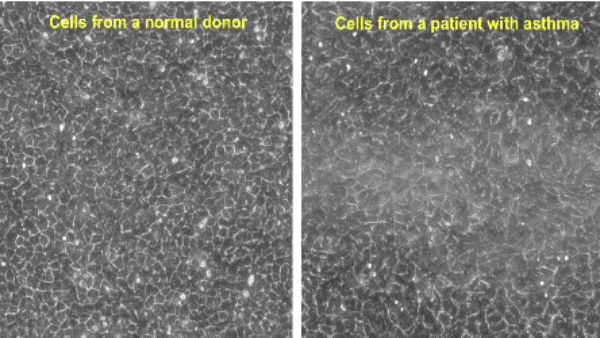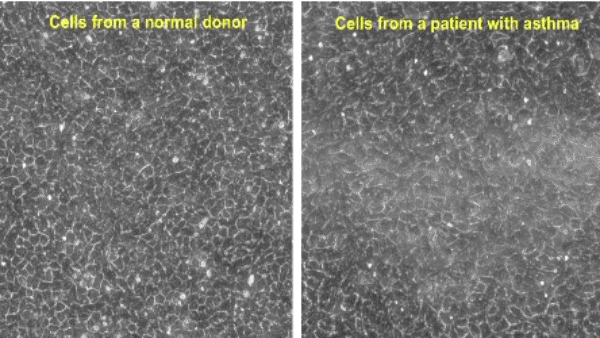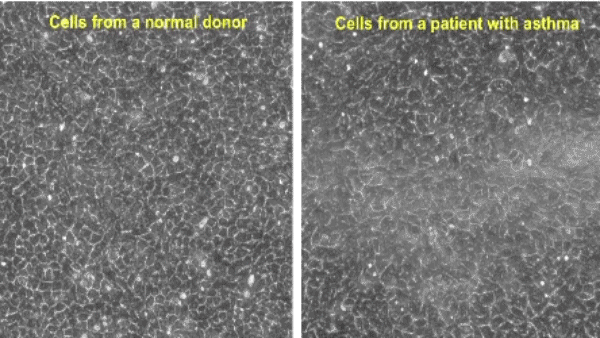
Research at the Park Lab is focused on understanding the role of the airway epithelium in the lung. As the primary barrier against external stimuli—ranging from environmental pollutants and allergens to bacteria and viruses—the airway epithelium serves as the first line of defense for respiratory health.
665 Huntington Avenue
Building 1, Room 306
Boston, MA 02115
Our research interest lies in understanding the role of the protective tissue lining the lungs, known as the airway epithelium. This tissue is the first line of defense for respiratory health, and serves as the primary barrier against external stimuli—ranging from environmental pollutants and allergens to bacteria and viruses—in the lungs.
While airway epithelial cells contribute significantly to lung homeostasis under normal conditions, their defense mechanisms are dysregulated under chronic conditions such as chronic obstructive pulmonary disease (COPD) and asthma. Learn more.
Who We Are
Research Opportunities
Two PhD programs are available to students who are looking for research opportunities in the Park Lab:
Population Health Sciences (PHS)
The PhD in PHS involves understanding origins and determinants of health and disease across populations. Students will choose a specific field of study from the following: Environmental Health, Epidemiology, Global Health and Population, Nutrition, Social and Behavioral Sciences. The main goal of this program is to provide students with the tools to shape new ideas in population health sciences, to communicate those ideas effectively, and to serve changing health needs in different societies and contexts.
Biological Sciences in Public Health (BPH)
The PhD in BPH involves understanding, preventing, and treating diseases affecting large populations. Students will obtain knowledge of both mechanistic and quantitative approaches to biomedical research, focusing on the metabolic basis of health and disease, immunology and infectious disease, gene-environment interactions, and inflammatory and stress responses. The main goal of this program is to provide students with a solid biomedical foundation for the application in interdisciplinary disease research.
Park lab has an immediate opening for postdoctoral fellow positions in the Molecular and Integrative Physiological Sciences program at the Harvard T.H. Chan School of Public Health, in Boston.
Applicants must hold a recent Ph.D. or M.D. in Biomedical Engineering, Molecular Biology, Cell Biology, Biochemistry or a related field. Previous experience in lung biology and mouse models of asthma is desirable.
Up to 3 years of support is available. Support from an NIH T32 training grant is available in the MIPS program.
To apply, please submit a letter of interest including your research goals, CV, and the names, email addresses, and phone numbers of three references to hsphparklab@gmail.com.